Horizon scanning projects
Horizon scanning projects are part of TLV's horizon scanning work to identify new and emerging medical technology that may become important in Swedish healthcare. They focus on a specified theme and are performed under a period of 2-3 months.
 Zoom image
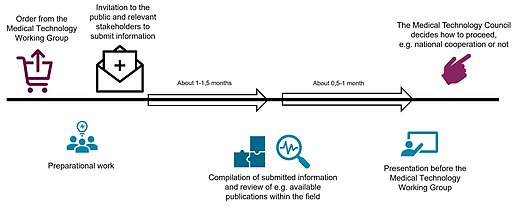
Zoom imagePicture shows the process of horizon scanning projects. Click to zoom in.
In a horizon scanning project, TLV invites stakeholders such as industry organizations, professional associations, patient associations, innovation offices at universities and other relevant authorities to inform TLV about new and emerging medical technologythat may be of interest for further evaluation at national level. The theme is typically selected by the knowledge management networks within the SKR, including national experts in each field of the healthcare system, together with the Medical Technology Working Group (beredningsgruppen).
The stakeholders are invited to submit information to TLV for a certain period (approximately 1-2 month(s)). The information should generally include the following:
- Name of medical deviceand company name
- Description of the intended use of the medical device
- Information about the medical device is CE-marked and/or FDA-approved
- Brief summary of clinical data and/or health data, if available
- Information about use of the medical device in Sweden today
- Link to website with additional product information
In a horizon scanning project, the information submitted to TLV is compiled and reported to the Medical Technology Working Group. The Medical Technology Working Group forwards interesting findings to the Medical Technology Council (MTP-rådet), which subsequently decides if there are any products to be further evaluated and considered for national recommendations.
See ongoing and completed horizon scanning projects under "Ongoing/Completed horizon scanning projects".
Page information
- Last updated
- 1 August 2022